Overview of Theme 2-2
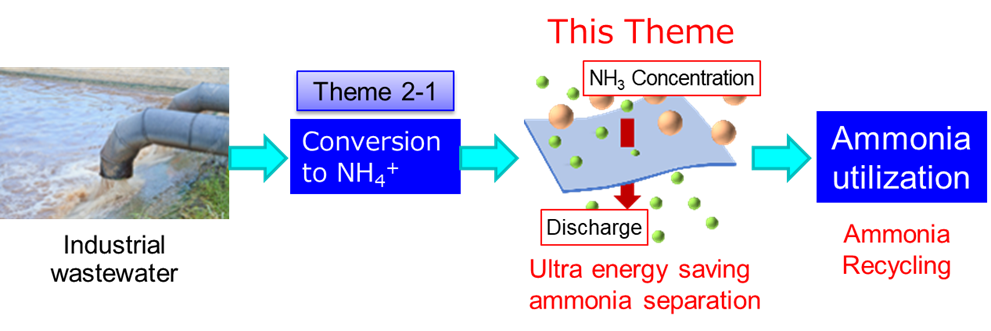
In Theme 2-2, we develop processes for the separation and concentration of NH4+ using several types of membranes and adsorbents with high performance to achieve a highly concentrated and widely applicable NH3 solution using the NH4+ solution converted in Theme 2-1. Innovative membranes and adsorbents must be developed to establish concentration processes with extremely low energy consumptions. The key point of this development is the reduction of energy consumption to the utmost limit.
Separation and concentration technology with extremely low energy consumption
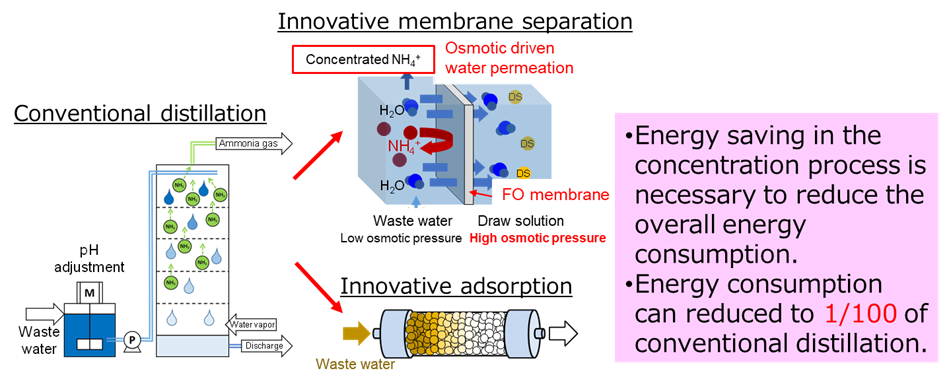
To date, distillation has been used for ammonia recovery from wastewater in some cases. The innovative membrane and adsorption processes targeted in this project exhibit a higher reduction in energy consumption compared to that exhibited by the conventional method. One example of an innovative membrane is the forward osmosis (FO) membrane. In the FO process, spontaneous water permeation occurs owing to the osmotic pressure difference, which enables the FO process to have relatively low energy consumption. In the adsorption process, an innovative adsorbent (Prussian-blue-type complex) with selective NH4+ adsorption properties, even in the presence of several ions, is developed.
Concentration process
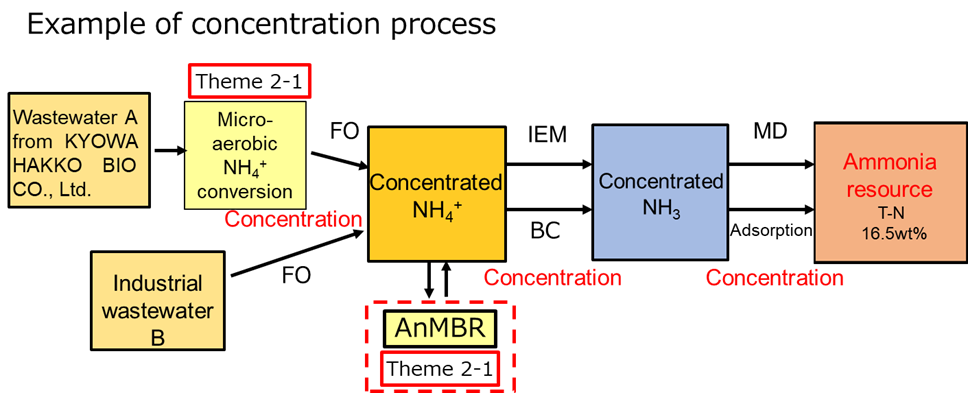
Examples of NH4+ concentration processes in industrial wastewater are shown in the below figure. In the case of wastewater A, after micro-aerobic conversion, NH4+ concentration is conducted by the FO process, followed by the addition of a brine concentration (BC) or ion exchange membrane (IEM). Finally, ammonia can be obtained via membrane distillation (MD) or adsorption. In the case of wastewater B, NH4+ conversion and energy production (methane gas formation) occur in the anaerobic membrane bioreactor (AnMBR) after the wastewater concentration using the FO process. The following concentration methods are the same as those described above.